Quality Assurance Officer
บริษัท โอลิค (ประเทศไทย) จำกัดThis position is assigned for handling of control raw material specification and procedure of QC part according to the established material specification and procedure also required update document related with raw materials part for registration.
Responsibility:
- Verify raw materials specification for new materials and update specification and test method for the current materials
- Prepare official raw material specification and analytical method.
- Prepare raw material control record.
- Provide the dossier to regulatory submission and responsible for the regulatory query on raw material specification.
- Update raw material specification to correspond with updated compendial pharmacopoeia.
- Bachelor’s degree in Chemical Science.
- Experience in Quality Control or Laboratory in Pharmaceutical industry will be advantage.
- Good command in English and computer literacy.
- Strong interpersonal and communication skill, Analytical skill.
- Persuasive abilities, Hands-on in all aspect of work.
- High responsible with the timeline commitment, Patient and positive thinking.
- Strong analytical and problem solving abilities, High responsible with timeline commitment.
- Dental insurance
- Free shuttle bus
- Life insurance
- กองทุนสำรองเลี้ยงชีพ
- การฝึกอบรมและพัฒนาพนักงาน
- ค่าทำงานล่วงเวลา
- ค่าทุนการศึกษา
- ทำงานสัปดาห์ละ 5 วัน
- ประกันสังคม
- ประกันสุขภาพ
- ลาบวช
 บริษัท โอลิค (ประเทศไทย) จำกัด
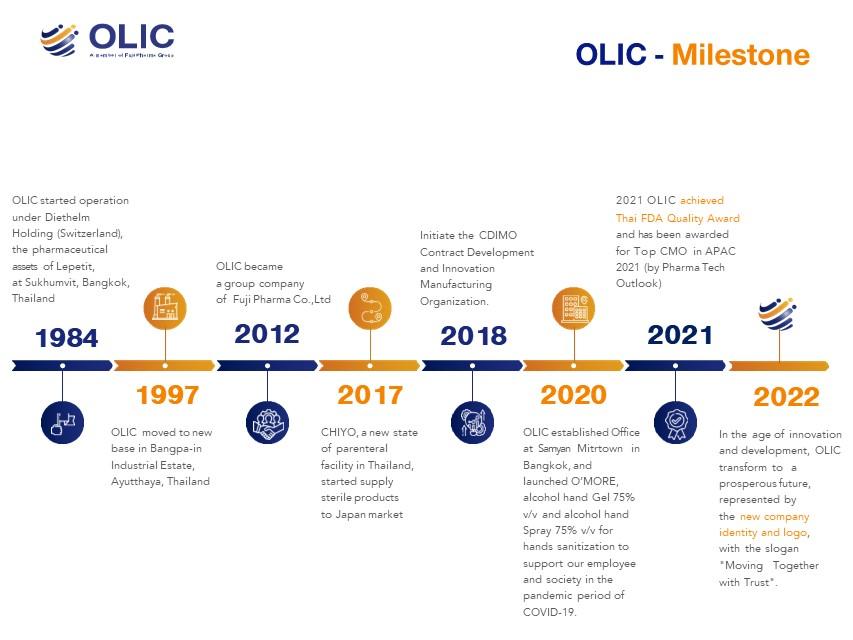
บริษัท โอลิค (ประเทศไทย) จำกัดToll and contract manufacturer of choice for Global Pharmaceutical, Healthcare and Personal Care companies.
OLIC has cultivated successful partnerships that last.
The company is a trusted partner of more than 30 multinational companies, including most of the Big Pharma organizations. The OLIC Manufacturing facility complies to GMP requirements to the PIC/S guide for Good Manufacturing Practice for Medicinal products. Products made by OLIC are serving not only markets in Thailand and South-East Asia, but also Japan, Australia, US, and Europe.